|
5/21/2023 0 Comments Polyatomic ion bonding memeIn some cases, three covalent bonds can be formed between two atoms. Similarly, in carbon dioxide (CO 2), two double bonds are formed between the carbon and each of the two oxygen atoms (Fig. For example, in the formation of the oxygen molecule, each atom of oxygen forms two bonds to the other oxygen atom, producing the molecule O 2. Sometimes two covalent bonds are formed between two atoms by each atom sharing two electrons, for a total of four shared electrons. Figure 2.30 A has additional examples of single bonds. For example, water, H 2O, has two single bonds, one between each hydrogen atom and the oxygen atom (Fig. A molecule can have multiple single bonds. A single bond is defined as one covalent bond, or two shared electrons, between two atoms. In the formation of a covalent hydrogen molecule, therefore, each hydrogen atom forms a single bond, producing a molecule with the formula H 2. 
Hydrogen is shown in Fig 2.28 with one electron. When an electron, or dot, from one element is paired with an electron, or dot, from another element, this makes a bond, which is represented by a line (Fig. The oxygen atom forms two bonds, one with each of two hydrogen atoms therefore, the formula for water is H 2O. The single electrons match up to make pairs (Fig. One oxygen atom can share its unpaired electrons with two hydrogen atoms, each of which need only one additional electron. To fill its valence shell, oxygen needs two additional electrons, and hydrogen needs one. 
In the formation of water, an oxygen atom has two unpaired electrons, and each hydrogen atom has one (Fig. First, determine how many atoms of each element are needed to satisfy the octet rule for each atom. 2.29 gives an example of how to make a Lewis dot structure. In order to form a covalent bond, each element has to share one unpaired electron.įig. The number of bonds that each element is able to form is usually equal to the number of unpaired electrons. The first four electrons are placed as single electrons, then the remaining four are paired. There can be up to eight dots, for eight valence electrons. 2.28 Dots are placed around the symbol of the element to represent the number of valence electrons in the element. A table of Lewis dot symbols of nonmetal elements that form covalent bonds is shown in Fig. Lewis dot structures are one way to represent how atoms form covalent bonds. It takes two electrons to make a covalent bond, one from each bonding atom. Covalent bonding is the type of bond that holds together the atoms within a polyatomic ion. Covalent bonding generally happens between nonmetals. This is a covalent bond, a bond in which atoms share electrons. This is how hydrogen and oxygen share electrons they each have an electron that they can share in a bond. The bones represent one of their electrons. Heating a hydrate can remove some or all of the water molecules from the compound.Imagine two puppies, each with a bone (Fig. A hydrate is named using a Greek prefix to indicate the number of water molecules per unit.Ī hydrate contains a specific number of water molecules per formula unit of compound. 
H3PO4 Which of the following statements correctly describe hydrates? Select all that apply. What is the name of the oxoacid with the formula HBrO3? bromic acid Select all the polyprotic acids. nitric acid nitrous acid The BrO3- ion is named bromate. The oxoacid of the nitrate ion is called _ while that of the nitrite ion is called _. An oxoanion containing two fewer oxygen atoms than the -ate ion is called the e ion.Īn oxoanion containing one more oxygen atom than the -ate ion will have the prefix per- added. Blank 1: monoproticīlank 2: polyprotic or diprotic Select the statements that correctly describe naming an oxoanion. NaNO3 An acid that has only one ionizable proton is termed _, whereas an acid with two or more ionizable protons is termed _. Which of the following formulas for ionic compounds are written correctly? Select all that apply. 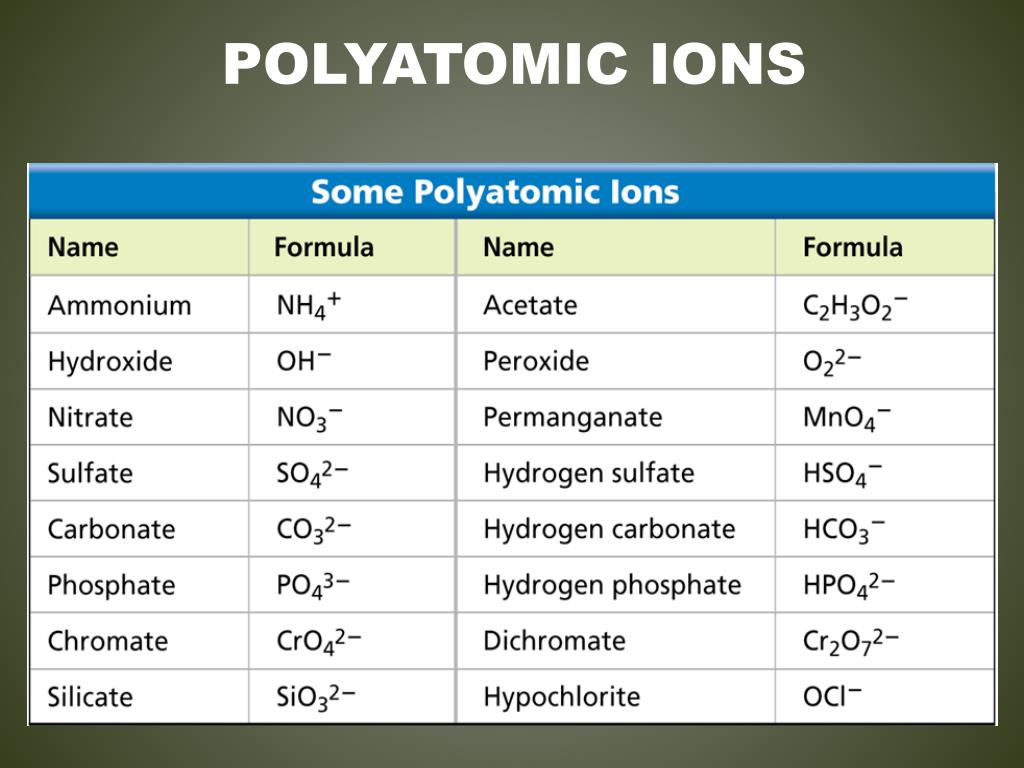
The acid derived from the bromate anion would be called bromic acid. The names of oxoacids are related to the names of the oxoanions from which they are derived. The acid derived from the chlorite anion would be called chlorous acid. polyatomic ionic What is the correct systematic name for the compound Na2CO3? sodium carbonate Which of the following statements correctly describe the rules for naming oxoacids? Select all that apply. A unit that contains two or more atoms covalently bonded together but that has an overall charge is called a(n) _ ion. The atoms of a polyatomic ion are held together by covalent bonds. The charge on a polyatomic ion belongs to the whole unit. Which of the following options correctly describe a polyatomic ion? Select all that apply.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
